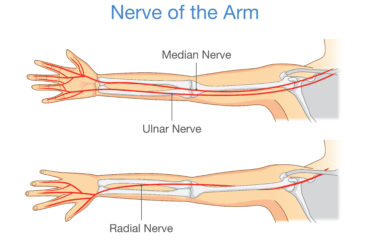
Stem cell and PRP procedures for rotator cuff bursitis are counted among the most advanced medical treatment modes. It prevents pain and disability from taking over your rotator cuff while initiating natural healing. An alternative to surgery, stem cell therapy for rotator cuff bursitis is minimally invasive and facilitates faster recovery.
What are stem cells?
Stem cells are those cells of our body that have the capability of renewing themselves as well as replicate other cells. These cells give birth to progenitor cells while renewing themselves. The second-generation progenitor cells are able to convert themselves into various other cells found in the human body.
What is rotator cuff bursitis?
Rotator cuff bursitis occurs when the bursa or small sac of fluid gets inflamed due to arthritis, injury, or wear and tear. The rotator cuff has four muscles that keep the arm attached to the shoulder. The bursa acts as a cushion between the muscle tendons and bones. When it gets inflamed due to injury, rheumatoid arthritis, degenerative disorders, overuse, or stressful occupation
What are various stem cell therapies used to treat rotator cuff bursitis?
Patients can for either stem cell injections or prolotherapy to treat rotator cuff bursitis. Both procedures have equal benefit and they only differ in the degree of treatment. Stem cell therapy is one-time procedure while PRP may need to be repeated more than one with a gap of 4 to 6 weeks.
How does stem cell therapy treat rotator cuff bursitis?
Stem cells create new cells called progenitor cells, which have the capability to become any type of cell found in the human body. In rotator cuff, these cells convert themselves into bursa cells and replace the old and damaged cells. This results in the regeneration of the damaged bursa and patients overcome pain and disability associated with rotator cuff bursitis.
Where from stem cells are obtained?
Two types of stem cells – mesenchymal and hematopoietic – are ideal for repairing and regeneration of the damaged rotator cuff bursa. These stem cells can be easily obtained from the bone marrow. Another rich source of these stem cells is the umbilical cord. However, very few people have the opportunity to preserve their umbilical cord at the time of birth leaving bone marrow the most preferred source.
Why stem cell therapy for rotator cuff bursitis is better that other modes of treatment?
- It is similar to the process of natural healing.
- Stem cell therapy has no side effects.
- It is non-invasive and performed in an outpatient setting.
- The recovery is quick and the healing is complete.
How is the stem cell therapy for rotator cuff bursitis performed?
The procedure is performed in two stages.
The first stage involves harvesting of stem cells. A needle in injected in to the iliac crest in the hip area. The skin above is numbed to avoid any discomfort. Multiple samples are obtained from the bone marrow. These samples collected are further refined in the lab using specialized methods and stem cells are separated from other cells. These stem cells are grown further for 1 to 3 weeks.
The second stage involves injecting of stem cells to the damaged rotator cuff. Doctors perform the procedure using general or local anesthesia. The stem cells are collected in a syringe and injected into the damaged rotator cuff under fluoroscopic guidance.
How successful is stem cell therapy for rotator cuff bursitis?
- A 2015 study reported by the World Journal of Stem Cells claims that stem cell procedures for rotator cuff bursitis have “better healing properties, ability to improve the amount of fibrocartilage formation, and increase in biomechanical strength of the shoulder.”
- A 2014 research report highlighted the growth and bioactive healing factors promoted by the stem cell therapy and found bone marrow mesenchymal stem cells as an improved way to ensure fast healing.
- Another 2014 study in the Tissue Engineering journal also admired the healing and improvements linked to stem cell procedures for rotator cuff bursitis.
Should I have any post-operative care?
You may have to use a sling for 2/3 days and keep the shoulder static. Sleep in a comfortable position. Doctors also advise patients to avoid stress on the shoulder for three weeks.
Do I need multiple stem cell procedures for rotator cuff bursitis?
When bone marrow-derived stem cells are used in the injection, the procedure is one-tome affair. In case of PRP, a patient may need more than one treatments with a 4-week gap in between.
How soon will I feel the improvement?
The initial improvements are felt within one to two weeks. However, the healing continues for months and the final result may take up to a year to become visible.
Who should not undergo stem cell therapy for rotator cuff bursitis?
The procedure is not suitable for those who have active infections and are undergoing cancer treatment. Those taking blood thinners or have complex medical issues should also stay away.
What are side effects of stem cell therapy for rotator cuff bursitis?
Stem cell procedures are safe for patients suffering from rotator cuff bursitis. Usually, the treatment is followed by temporary fever and injection-site soreness for 24 to 48 hours. These problems resolve without any complication.
References
Mora MV, Ibán MAR, Heredia JD, Laakso RB, Cuéllar R, Arranz MG. Stem cell therapy in the management of shoulder rotator cuff disorders. World Journal of Stem Cells. 2015;7(4):691-699.
Randelli P, Randelli F, Ragone V, et al. Regenerative Medicine in Rotator Cuff Injuries. BioMed Research International. 2014;2014:129515.
Song N, Armstrong AD, Li F, Ouyang H, Niyibizi C. Multipotent mesenchymal stem cells from human subacromial bursa: potential for cell based tendon tissue engineering. Tissue Eng Part A. 2014 Jan ;20(1-2):239-49.
Steinert AF, Kunz M, Prager P, et al. Characterization of bursa subacromialis-derived mesenchymal stem cells. Stem Cell Research & Therapy. 2015;6(1):114.